Medtronic wins FDA clearance for robot in cranial, ENT surgeries
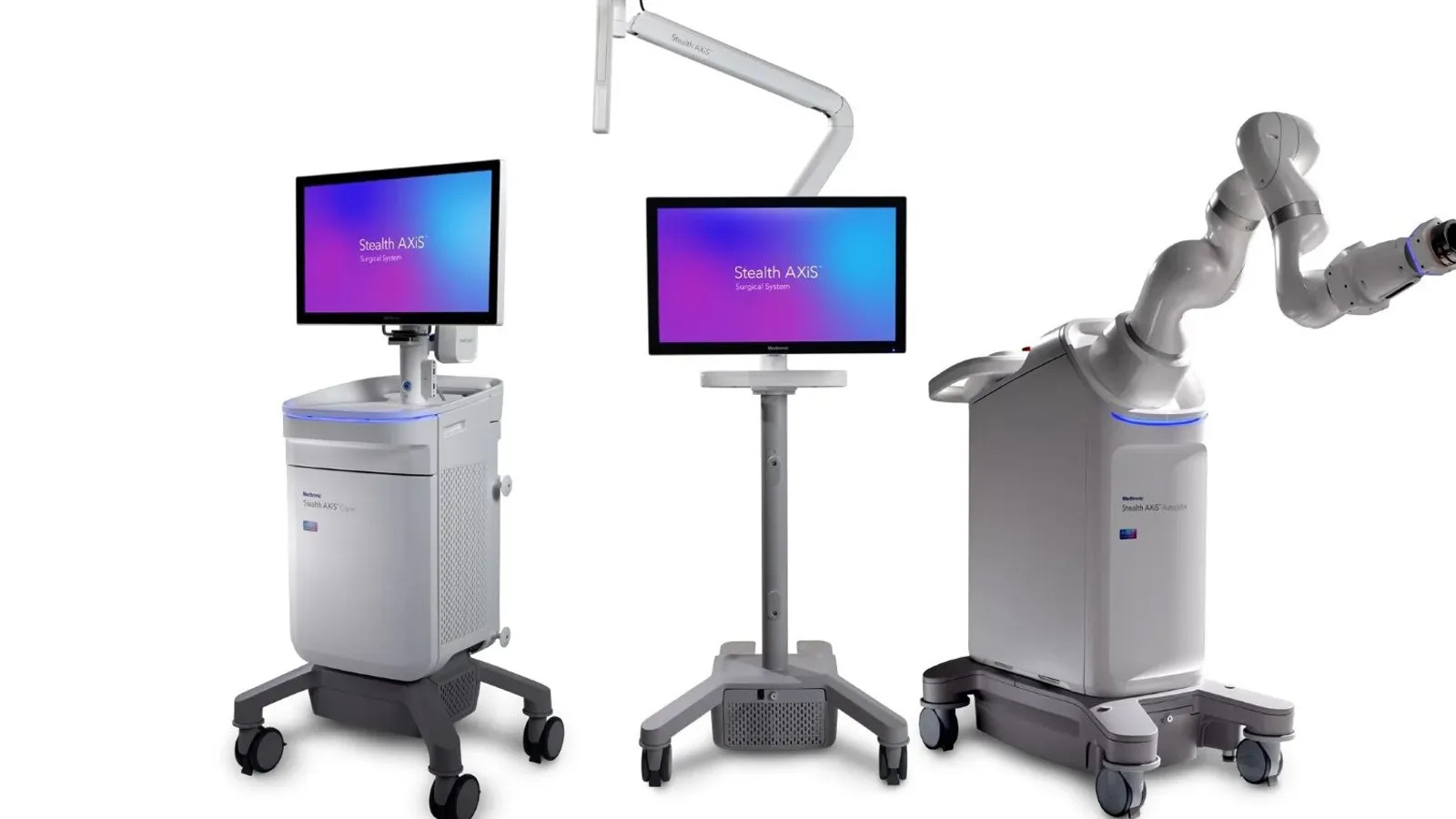
The surgical navigation and robotics system is part of the company’s push to expand its share of the $15 billion cranial and spinal technologies market.
The Food and Drug Administration cleared Stealth AXiS for use in spine procedures in February. At the time, Medtronic said the underlying architecture could support cranial applications and ENT surgeries, pending 510(k) clearance. The FDA cleared the system in ENT procedures on March 16 and followed up with a 510(k) nod in cranial applications on March 26.
Michael Ivan, neurosurgeon at the University of Miami Health System, said in a Medtronic statement that physicians make better decisions in complex brain surgeries when they have more information. Ivan added that preoperative planning with tools like tractography, a 3D modeling technique, may support better patient outcomes.
Stealth AXiS uses artificial intelligence-enabled tractography to map the brain. Medtronic has integrated the system with GE HealthCare’s bkActiv to provide real-time ultrasound imaging during procedures.
Medtronic said its ENT system offers navigation and visualization tailored to the anatomy of the sinuses and skull base. Claimed benefits include sharper anatomical detail and faster responsiveness to support confident decision-making in the operating room.
Vijay Ramakrishnan, professor of otolaryngology-head and neck surgery at Indiana University School of Medicine, said in a statement that intraoperative navigation is critical in minimally invasive endoscopic surgery of the sinuses and skull base. Navigation helps surgeons “delineate complex anatomy within high-risk spaces,” Ramakrishnan said.
Medtronic CFO Thierry Piéton said on a February earnings call that he expected the Stealth AXiS spine product to add to sales in the current quarter. The system is part of the company’s push to expand its share of the $15 billion cranial and spinal technologies market.