Monthly Archives: March 2026
-
- March 31, 2026 - 1 min read
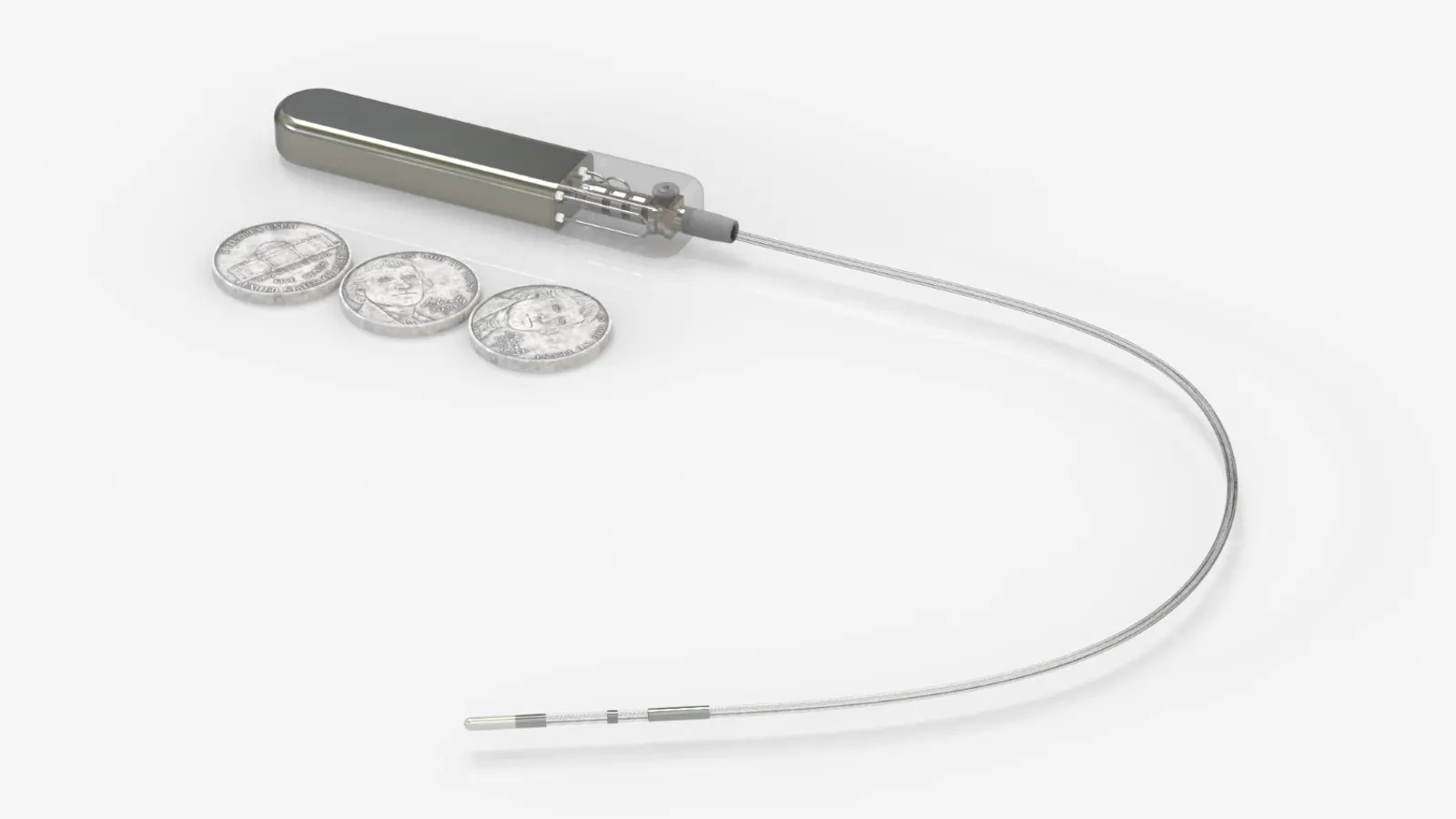
Glucotrack plans to file for an investigational device exemption in the second quarter, with hopes of starting a trial in the second half of 2026.
Glucotrack said it is ready to start a U.S. clinical trial following progress in other countries. Last year, the company shared results of a first-in-human trial of the device in Brazil.
During the five-day trial, the 10 participants had no procedure- or device-related adverse events, and the device demonstrated a mean absolute relative difference, or
-
- March 31, 2026 - 2 min read
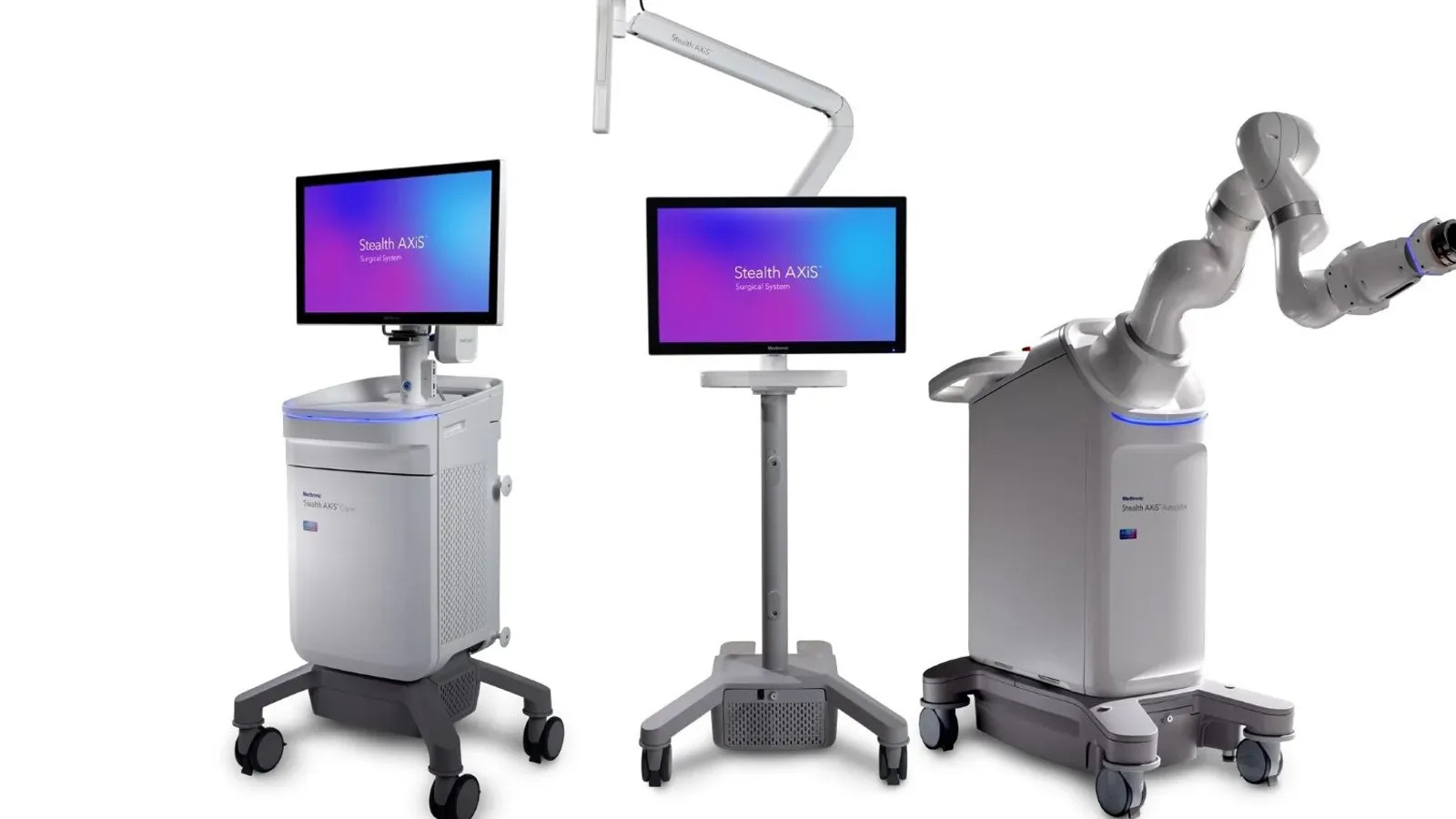
The surgical navigation and robotics system is part of the company’s push to expand its share of the $15 billion cranial and spinal technologies market.
The Food and Drug Administration cleared Stealth AXiS for use in spine procedures in February. At the time, Medtronic said the underlying architecture could support cranial applications and ENT surgeries, pending 510(k) clearance. The FDA cleared the system in ENT procedures on March 16 and followed up with a 510(k) nod in cranial applications on
-
- March 31, 2026 - 2 min read
The company said it will seek an expanded indication for the left atrial appendage closure device after it met all safety and efficacy goals in the closely watched CHAMPION-AF study.
The Watchman device is currently approved by the Food and Drug Administration for use in patients with atrial fibrillation who cannot take long-term anticoagulants due to a high risk of bleeding.
The implant permanently closes off the left atrial appendage, a small pouch where blood can pool and form clots that can travel
-
- March 31, 2026 - 2 min read
Medtronic detailed plans to further study the blood pressure treatment while releasing additional data from the SPYRAL HTN trials that showed the procedure reduced hypertensive emergencies.
Medtronic’s renal denervation system delivers radiofrequency energy to ablate nerves around the renal arteries, disrupting overactive signaling between the kidneys and the brain to reduce blood pressure.
In October, the Centers for Medicare and Medicaid Services granted a national coverage determination for renal