Medtronic to study renal denervation combined with PCI

Medtronic detailed plans to further study the blood pressure treatment while releasing additional data from the SPYRAL HTN trials that showed the procedure reduced hypertensive emergencies.
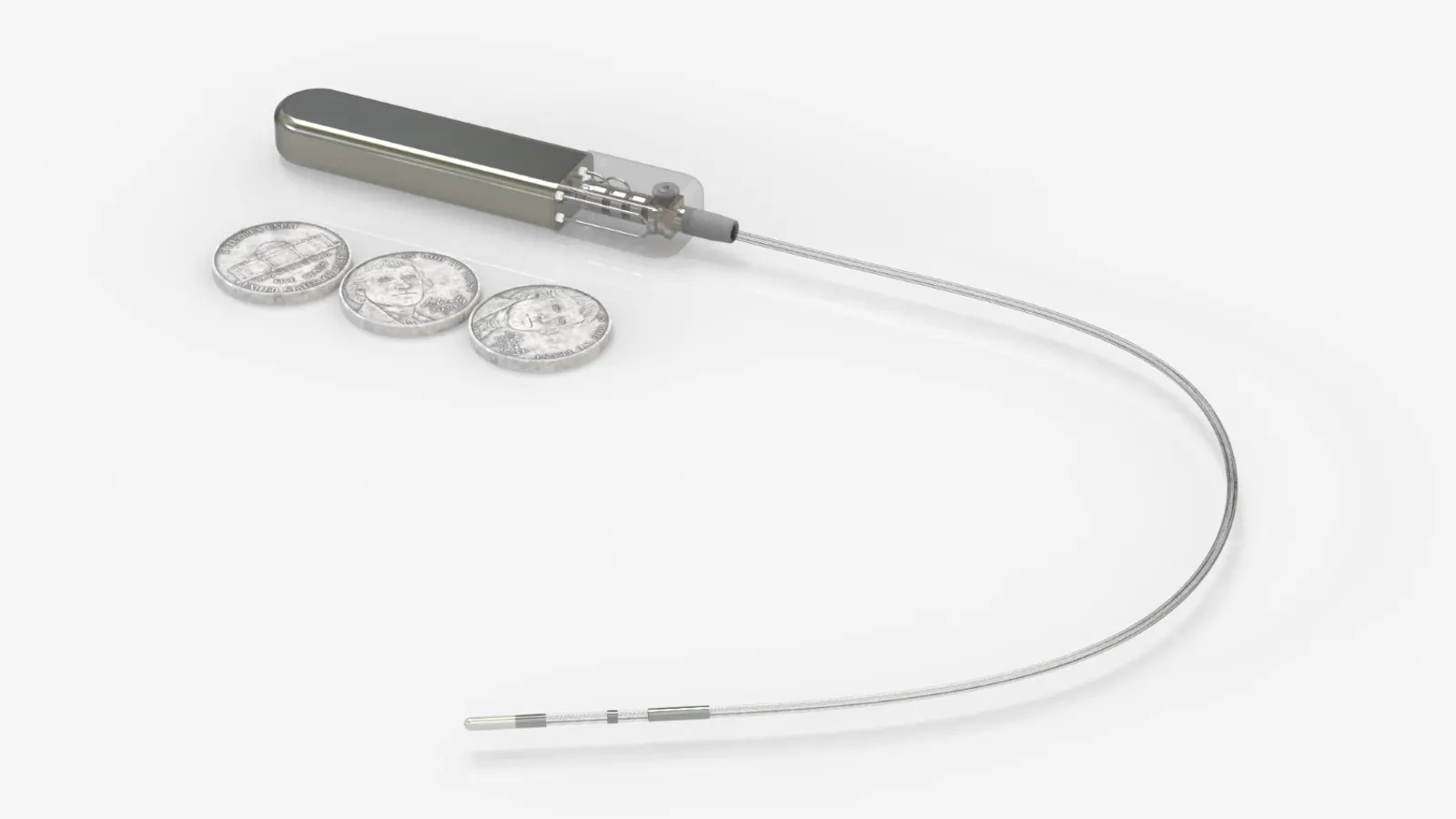
Medtronic’s renal denervation system delivers radiofrequency energy to ablate nerves around the renal arteries, disrupting overactive signaling between the kidneys and the brain to reduce blood pressure.
In October, the Centers for Medicare and Medicaid Services granted a national coverage determination for renal denervation, paving the way for increased use of the technology to fill a treatment gap for patients when lifestyle changes and prescription medications have failed to lower their blood pressure.
Medtronic’s randomized, multicenter EMBRACE trial will enroll 1,000 patients to compare renal denervation combined with PCI to PCI alone. Medtronic is interested in understanding if the approach will improve outcomes in high cardiovascular risk patients, said Jason Fontana, general manager of Medtronic’s renal denervation business.
The study’s composite endpoint will include cardiovascular death, stroke, heart attack and hospitalization for heart failure or hypertensive crisis at two years.
Roxana Mehran, a professor at the Icahn School of Medicine at Mount Sinai who will help lead the study, said the research will address residual cardiovascular risk after PCI.
“Uncontrolled hypertension remains a key driver of recurrent events,” said Mehran, “and EMBRACE explores a potential ‘sweet spot’ where revascularization and renal denervation can be combined to move from intervention to prevention.”
In the SPYRAL HTN pooled analysis, patients treated with renal denervation had a statistically significant lower medication burden at three years, according to Medtronic. In addition, the company said reductions in emergency department admissions for hypertensive events could save an estimated $380 million a year in U.S. healthcare costs, assuming a treatment adoption rate of 50%.